The immune system is the body’s defense mechanism to protect itself from harm. This harm can come from outside the body in the form of bacteria and viruses, and from inside the body, when cells mutate or change to become cancerous. Our immune system is very complex and is made up of many different types of cells, all of which work in different ways to protect the body.
An example of one type of immune system cell is the cytotoxic T cell, also known as T lymphocyte, which acts to protect the body by producing a chemical that causes foreign cells to kill themselves.
In recent years immunotherapies have emerged as a way to to harness your own body to defeat cancer and generate a long-lasting response. Most of these therapies involve unleashing the T Cells to attack cancer cells. For certain cancers there have been great results and early successes.
But while there have been great leaps forward, this powerful approach has only worked in 20-30% of patients. Although the immune system is more than capable of destroying cancer cells, it avoids targeting the body’s own cells. Unfortunately, tumours have developed ways to exploit the mechanisms that immune system has for avoiding killing healthy cells, and to get this powerful new form of treatment working for more cancer patients we must overcome the tumours’ manipulation of our immune system.
Tumour Environments
One-way cancer cells manage to keep safe from the immune system is by hiding in the collagen rich environment of a tumour. Collagen is like a scaffolding for the body, it is an important structural protein in organs, bones and skin. While collagen is important for keeping your body structurally sound, tissues that are particularly collagen dense have been shown to inhibit the movement of T cells (1). Tumours can also contain collagen. As mentioned earlier, T cells can produce a chemical which causes foreign cells to kill themselves. However, even if the T cell knows where to go and what it’s looking for, it might not be able to get close enough to the cancer cells to induce their death because it gets lost in the collagen of the tumour.
Tumours are also sites of rapid cell growth. With rapid cell growth comes the need for many nutrients and building blocks from which new cells are formed. One of the characteristics of cancer cells is that they grow uncontrollably and consequently will take a significant portion of the nutrients and building blocks available in the area of the tumour leaving none for the immune system’s cells. For example, glucose, one of the main sugars a cell needs for energy to grow, is also critical for activating cancer fighting T cells. If a T cell arrives at a tumour, a lack of glucose means that it cannot do its job. The cancer cell is then safe.
Immune System Inhibition
The effectiveness of the immune system is determined by its ability to destroy harmful cells. Macrophages can engulf and digest cancer cells. However, some cancers can stop macrophages from killing them. For example. leukaemia, a cancer of the body’s blood-forming tissues cells, has an antigen called CD47 on their surface. This antigen, CD47 interacts with a protein found on macrophages and basically tells the macrophage to stop eating.
Melanoma cells, a cancer of the skin, do a similar thing to cytotoxic T cells. These cancer cells release soluble protein, PD-L1 into the bloodstream from their surface. If soluble PD-L1 proteins contacts a cytotoxic T cell, they act as an order to stop the T cells from multiplying. The T cells may know what to attack and successfully destroy a few cancer cells, but their numbers will be too few to make a difference. Scientists at the University of Pennsylvania noticed this and subsequently did research on the effects of using a molecule to block the receptors on T cell to which the PD-L1 proteins bind. They hoped that by blocking this interaction, then there would then be enough T cells made to kill the cancer cells.
Here’s the bad news: ovarian carcinoma tumour cells have been found to produce a chemokine named CCL22 (4). A chemokine is a protein used to signal other cells to migrate or move somewhere else in the body. In this case, CCL22 is signaling to suppressor cells such as regulatory T cells (T-regs for short) to come over to the tumour. T-regs help the body’s natural suppression of the immune system after overcoming an illness. In effect, they turn off that part of the immune system after the work is done. But in cancer when T-regs arrive at a tumour, they suppress, or stop the body’s immune system cells from killing cancer cells. This is called immunosuppression. So the cancer cells produces a signal which cloaks it from the immune system. It is an incredibly deceptive and effective tactic used by cancer cells to tell the body there’s nothing going on here.
The Comeback: Immunotherapies
For every punch from the immune system, it seems that cancer cells return with an equally effective counterpunch. However, scientists have been effective at getting the immune system back on its feet. With the difficult side effects and sometimes low success rates of radiation and chemotherapy, immunotherapy may be a more effective way to treat cancer. There are several different approaches to immunotherapy, including using monoclonal antibodies, checkpoint inhibition, and oncylitic viruses.
Monoclonal Antibodies
Countless antibodies flow through the bloodstream and tissues of your body, tagging bad or foreign cells for the immune system to target. One way to use the immune system to kill get cancer cells is to make antibodies to cancer cells outside of the body, in the lab. Once scientists isolate a cell that produces an antibody that binds to cancer cell antigens, they clone the cell so that they can create large amounts of that specific antibody to administer to the patient. “Monoclonal” antibodies simply means that these antibodies are from a clone of one cell producing the appropriate antibodies. These cancer specific antibodies can then be given back to patients as a medication and they can provide an important jumpstart for a cancer patient’s immune system.
There have been approved drugs using monoclonal antibodies for several years now, including Rituximab which targets lymphoma and leukaemia cells. One limit of monoclonal antibody therapy is that researchers have found it difficult to target antigens presented by a wide array of cancers. Instead, monoclonal antibodies usually only target a specific type of cancer cell meaning a monoclonal antibody that can be used to treat leukaemia might not be effective for breast cancer patients and vice versa.
Checkpoint Inhibition
As mentioned above, some cancers can produce proteins which suppress the body’s immune system i.e. stops it from killing the cancer cells, or immunosuppression. Checkpoint inhibition is a specific type of monoclonal antibody therapy focused on reversing immunosuppression. Checkpoint proteins exist on immune system cells so that they can be stopped when the threat to the body, for example an infection, is under control, and the immune system response is no longer needed.
Cancer cells-produce proteins, like PD-L1 (mentioned above) and CTLA-4, which essentially activate brakes for the immune response. The goal for researchers is to find ways to stop this brake system used by cancer cells, so that immune system cells will ignore the suppression signals of cancer cells, and instead recognise and kill the cancer cells.
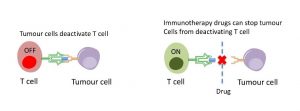
There are several researchers working on ways to block PD-1 (the checkpoint protein on T cells that cancer-produced PD-L1 targets). Nivolumab (trade name: Opdivo) is a medication that blocks cancer signals to PD-1. It has already been approved in some countries. Similarly, ipilimumab (trade name: Yervoy) targets CTLA-4, another checkpoint protein on T cells, and it is used to treat melanoma. Late last year, Bristol-Myers Squibb released clinical trial data for advanced melanoma patients treated with both Opdivo and Yervoy. They showed a 53% survival rate after four years. This is higher than the 20% survival rate for five years, which has been the average survival for this patient group according to the American Cancer Society.
Oncolytic Viruses
The first trials of any kind of immunotherapy were conducted by Dr. William B Coley when he injected bacteria into the tumours of patients way back in 1891. The intention behind this was to induce an inflammatory response at the site of the tumour, by alerting the immune system to the tumour. While some patients saw the remission of their cancer, other doctors didn’t think the risks were worth it and continued to look for solutions in surgery and radiotherapy.
A similar methodology has reemerged in the past few years. Viruses that specifically infect cancer cells can be used to invade tumours, subsequently multiplying, and causing the cancer cells to burst and reveal their mutated DNA to the body’s adaptive immune system (7). By lysing, or bursting the cancer cells, the virus will also stop the tumour’s overall growth and success.
TVEC, an oncolytic virus that can infect melanoma, or skin cancer cells, was approved in the US in 2015 (8). It is injected directly into melanoma tumours, killing cancer cells and encouraging the body’s immune system to identify and kill the tumour. TVEC is actually a type I herpes simplex virus engineered to replicate more effectively in tumour cells.
Of course, as with almost any other cancer treatment, there are risks. Intentionally putting a virus into your body could simply mean trading one disease for another.
However, the benefits may outweigh the risks. There have already been early stage clinical trials infecting brain tumours with viruses for positive results (9).
Conclusion
There is a long and arduous journey ahead in the fight against cancerous immunosuppression. There are bound to be discoveries of even more ways cancer can become invisible to the immune system. However, immunotherapies provide several new approaches to cure cancer, with even more possibilities being researched daily.









 Contact
Contact